 
Dilution factor is the amount of solvent required to reach a given concentration of solution for a given volume. Dilution is the process which involved making the solution weak or less concentrated. In serial dilution, the entire dilution factor will be multiple together to be the actual dilution factor. Serial dilution-agar plate technique counts only viable cells whereas other methods count both viable and dead cells. As example, if 1.0 ml of 3M HCl is added with 9 ml of distilled water, the dilution factor becomes 1/10. 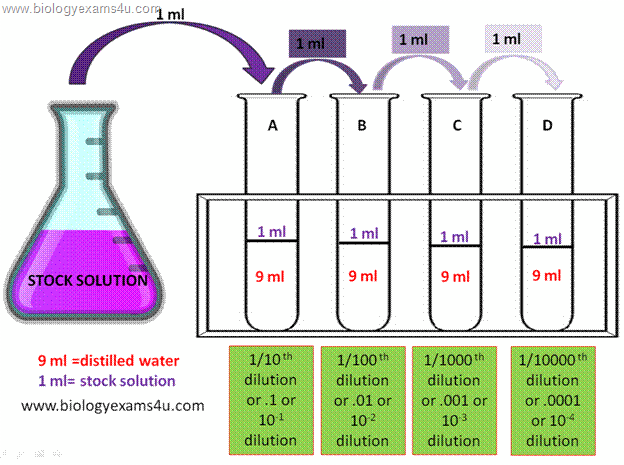
The factor can be obtained by dividing the actual volume of the initial chemical used to the final volume of the chemical after water is added. 
One way is by obtaining a factor called the Dilution Factor (DF). After performing the dilution, we need to know how much difference are the diluted chemical and the initial, undiluted ones. Thus, serial dilution simply means a series of repeated dilution performed on the same chemical basically to change its concentration. It is usually done if the chemical concentration is too high than the desired composition. Notice the color gradually getting paler as dilution advances.ĭilution is the act of mixing a chemical with other substance, usually distilled water to make it lighter in composition. Serial dilution performed on blue dye solution. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
January 2023
Categories |
 RSS Feed
RSS Feed
